Joule-Kelvin Expansion and the Inversion Curve
A casual walkthrough of Joule-Kelvin (Joule-Thomson) expansion and the throttling process — the much simpler way to cool a gas that won't give you cancer hehe.
lolll this one, I think you really have to read it rururururight in sequence with the previous post for the logic to be clear.
If I had written everything up to what I’m about to say now in the previous posting,
sluuuuuuuuuuurp~
it was going to end up like that, so I had no choice but to cut it and write it separately, please bear with me hehehe
Alright!!! So we’ve found one answer among the methods for cooling something down
namely, that doing Joule expansion works!!!!
So let’s say some professor assigns us homework like “go make the gases in the balloon a little cooler^^”
and since we know that ‘Joule expansion’ cools a gas,
we’re going to carry out the assignment with that principle.

Are you trying to give me cancer????
If I had to do that just to cool one little thing I think I’d get cancer T_T T_T T_T T_T T_T T_T
The model we want is
we want a simple model like this, right…..T_T T_T T_T
Now let’s study the simple model like the shape above.
They say the first step for such a simple model was taken by ‘Joule’ and ‘(William) Thomson’.
And later ‘Kelvin’ is said to have added to the achievement
So the expansion we’re going to study from now on
is the expansion called Joule-Thomson expansion, or
Joule-Kelvin expansion.
The schematic diagram is as follows.
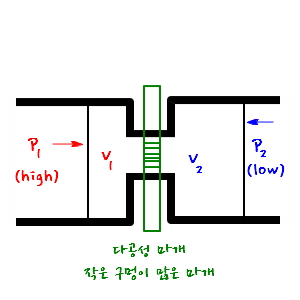
Yep@@@ that’s right. It really does look like shit, doesn’t it hehehe ha… he.
Anyway we gotta study letsgoletsgoletsgo
Let’s say gases at high pressure p_1 and volume V_1 flow into a place of low pressure p_2, and the volume becomes V_2, yep.
This kind of process is called a throthling process,
and such a throttling process has a characteristic.
As for what characteristic,
let the internal energy of the gas at V_1 be U_1
and the internal energy of the gas at V_2 be U_2.
As the gas at V_1 all moves over to V_2, the internal energy changes from U_1 to U_2.
That is, the amount of change in internal energy is
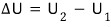
.
Where did this change in energy come from?!?!?!?!
It received work, and at the same time it did work, right??? This much of a feel
I’ll write out the work these guys ‘received’ and the work they ‘did’ and write the equation for the energy change.
At V_1 it received work of dW_1. (this would be the positive direction)
At V_2 it did work of dW_2 (this would be the negative direction)
that is
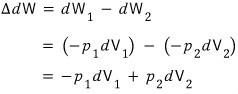
But, since this expansion is an expansion in which the pressures are maintained at p_1 and p_2 respectively,
eve~~~~rything goes over
that is,

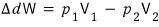
However, since it’s adiabatic,
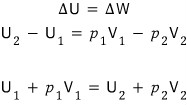
Eek!!!!! H = U + pV
Enthalpy appears?!?!?!?!
that is
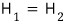
This is the throttling process
that is, the characteristic of the Joule-Kelvin expansion!!!!
So ’that kind of expansion’ we drew is automatically an expansion in which the enthalpy H is conserved
In other words, expanding while conserving H is what’s called Joule-Kelvin expansion!!!
(Conversely, expanding while conserving the internal energy U was Joule expansion)
Now let’s define the Joule-Kelvin expansion coefficient (Joule-Kelvin Coeifficient).
The Joule-Kelvin expansion coefficient is defined like this hehehe
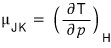
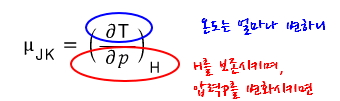
The meaning would be about this
But holding H constant and varying the pressure p doesn’t quite click.
So, again, I’m going to try to massage the expression using the reciprocal theorem.
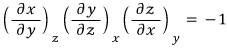
This expression….. again….link…. http://gdpresent.blog.me/220587333984

What I studied in thermal/statistical mechanics #23. Reciprocity theorem - reciprocal theorem, reciprocity theorem (recip…
As I mentioned in the previous posting, I’ll prove the reciprocity theorem! Of course it’ll be a link-only posting hehehe x is…
blog.naver.com
Anyway, so I’m going to plug in x = T, y = p, z = H like this
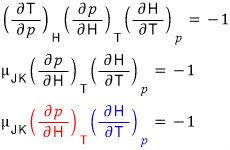
And now, using the definition of enthalpy H,
let’s try expressing it in state functions that are easier for us to handle
First!!!! When p is constant, what was the meaning of dH?!?!?!?
Yep, the meaning was dQ.

Gotta find the other one too

Alrighty sooo~~~ let’s finish up the Joule-Kelvin expansion coefficient over there.
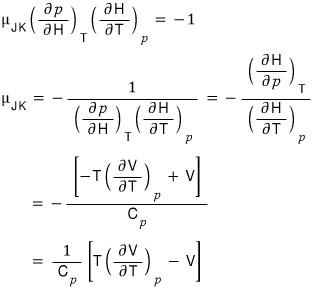
Then let’s think about it.
How much of a temperature change happens when you do a Joule-Kelvin expansion!
When we do a Joule-Kelvin expansion from pressure p_1 to p_2, I’ll write the temperature change that occurs like this

First let’s see how this amount works out for an ideal gas.
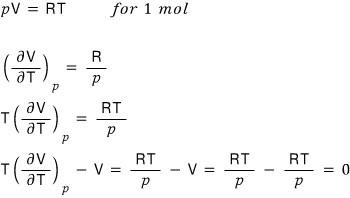
Ahaaa just like Joule expansion, for an ideal gas
we can see that even if you do a Joule-Kelvin expansion, no temperature change occurs, yep
Then let’s do it for the van der Waals gas too
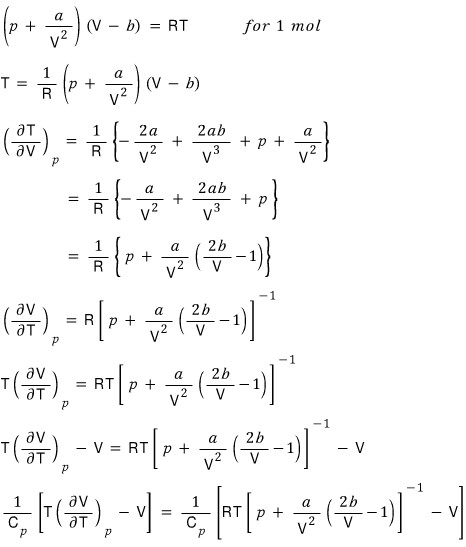
I’ll show it in a practice problem, but
the constant-pressure heat capacity of a van der Waals gas was computed like this.
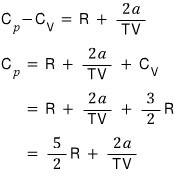
Therefore
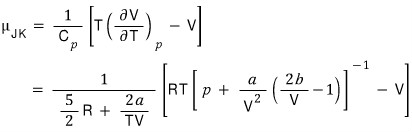
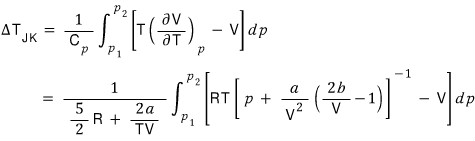
Wow….. it would’ve been great if we could figure out whether this is positive or negative now,
and even figure out how much positive, how much negative,
but we can’t calculate it, yep.,,T_T T_T T_T T_T how much cooling, how much heating..T_T T_T T_T?
For now, with the van der Waals gas model, it’s a fail.
Can’t say exactly.
So now I’ll turn my eyes to experimental physics.
Hmm, how values like that actually play out in reality
can be figured out in a different way.
First, the answer is that the value above can actually be “positive, or negative,,,” — that’s the answer.
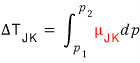
When that red blob in the middle is negative, the temperature change is positive. That is, it means it gets heated
when that red blob in the middle is positive, the temperature change is negative. That is, it means it gets cooled
Since it’s going from high pressure to low pressure, the sign flips the opposite way.
Alright then, what we’re going to think about now is
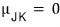
we’ll look for where this happens,
That part
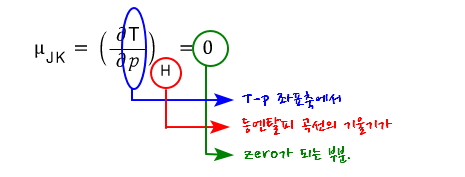
is found by this logic.
So first, let’s take a look at what isenthalpic curves look like on the T-p coordinate axis.
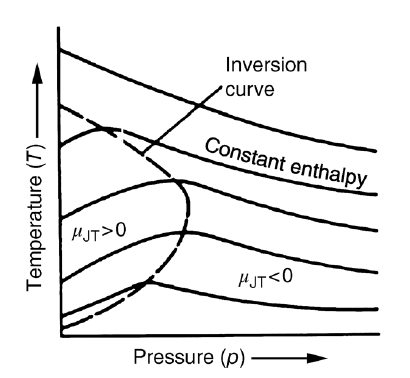
The isenthalpic curves for a real gas are said to look like this.
And in the graph above, the parts of those curves where the slope is 0 are all collected and connected by dashed lines.
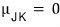
Yep that’s right. They say the plot of the set collecting only the p-T ordered pairs that make the Joule-Kelvin coefficient 0 is defined as the ‘inversion curve’.
Now then,
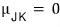
to the left of the part where this holds,
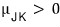
it is (meaning the slope is positive)
conversely, to the right
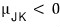
it would be. (the slope is negative)
When that coefficient is positive, we summarized that the temperature change due to Joule-Kelvin expansion is negative
that is, that it gets cooled
conversely, when that coefficient is negative, the temperature change due to Joule-Kelvin expansion is positive
that is, we summarized that it gets heated.
Therefore
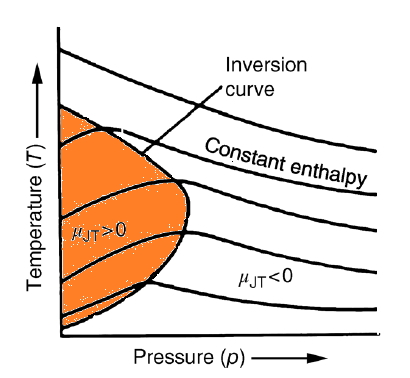
in the p-T states included in the colored region here, if you do a Joule-Kelvin expansion,
the temperature change due to expansion is… negative, meaning it gets cooled
and under (p,T) conditions other than that, doing a Joule-Kelvin expansion means it gets heated.
Another feature that’s visible
is that above the highest temperature on the inversion curve, it shows that you can’t heat it by doing a Joule-Kelvin expansion.
That is, to ‘cool’ something by Joule-Kelvin expansion, at the very least
it has to ‘already’ be in a state lower than ’that highest temperature’.
(That highest temperature is called the ‘inversion temperature’, they say)
So let’s say we were assigned the task of cooling He gas.
The inversion temperature of He is said to be about 45K.
So in order to cool helium by Joule-Kelvin expaaansion
first we need to have cooled it down to 45K by some other means……..hohohohoho
Liquefying helium is reeeeeeally difficult.
So liquid helium is reeeeeeeeeeeeally expensive, they say
So!!!!!!!!!!!! taking a single MRI scan costs an insane amount of money, they say
Because in order to operate an MRI machine once, you have to pour buckets of that expensive liquid helium, they say T_T T_T T_T T_T T_T T_T T_T
Oof.T_T.T_T T_T
Ah, and why is cooling to ultra-low temperatures so important,
because ultra-low temperatures are really necessary. Because at reeeally low temperatures you can discover really weird physical phenomena you normally couldn’t see,
for example superconductivity
When the temperature drops a lot, certain substances show superconductivity, right
If they show superconductivity, ….. if you utilize this it’s no joke, they say
you could even develop a train with a speed of about 3000km/h (if this gets made you can go to China for lunch and come back lolololololol)
And more than anything….. there’s no energy loss due to resistance, right,….????
So low-temperature physics is said to be a really important topic,
the most representative ‘superconductor research’ was enormously popular in the 70s and many scientists jumped into superconductor research, they say.
The result was ’total defeat’
They still don’t know, they say.
They don’t know anything, they say
That’s how ignorant humans are…..
Anyway lololololololololol cooling, this is just hopeless lolololololololololololololololol
I guess we shouldn’t cool stuff lolololololololololololololololololololololol
Originally written in Korean on my Naver blog (2016-07). Translated to English for gdpark.blog.