Trouton's Rule
A casual dive into Trouton's Rule, unpacking why phase transitions happen and how latent heat ties into entropy — because stability is literally everything.
The phase transition I kept foreshadowing from earlier posts — here we go.
Phase transition just means… well, the state of matter changes…. solid to liquid, liquid to gas, that sort of thing.
Why do phase transitions happen in nature??!?!
Everything seeks to be in a stable state, you know….
Which means, ’the phase-transitioned state is the more stable state (at that temperature and at that pressure)'
That is, at 150℃,
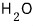
is more stable as water vapor (gas) than in any other state, and
at 50℃,
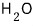
is more stable as water (liquid) than in any other state, and
at -50℃,
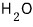
is more stable as ice (solid) than in any other state.
We learned this almost as an ‘axiom’ in middle and high school, and accepted it without any problem,
but now it’s time to look at it a little more rigorously from a thermodynamic point of view.
The first step will be latent heat.
When the state of matter changes, there’s an extra bit of thermal energy needed,
and we learned to call that ’latent heat’.
I think we should understand this as a physical quantity you can figure out experimentally or phenomenologically.
Aaaaand we also learned it as energy related to binding energy, right?!?!
So let me slap down the wiki definition of ’latent heat'.
Latent heat (潛熱) means hidden heat. It’s the heat released or absorbed when an object’s state changes without a change in temperature.
It’s heat generated by evaporation and condensation,
and it manifests as thermal energy going into water vapor when water evaporates from a water surface or from the surface of moist soil.
latent heat : L
extra heat needed to change to from phase 1 to phase 2 at constant temperature(T_c)
So, this latent heat thing we’re studying is about dQ, the heat quantity?!?!
Then, with the help of entropy — the physical quantity related to heat — let’s try to convert it into the other state functions we usually use.
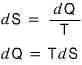
If we call the latent heat L, then the latent heat L is
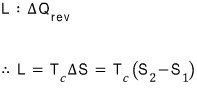
So, latent heat turned out to be related to the difference in entropy between the two phases at the critical temperature.
Since latent heat can only be discussed at the specific temperature where the phase transition occurs,
T isn’t a variable — we plug in the specific temperature T_c (the critical temperature).
Now let’s look at the T-S graph for water.
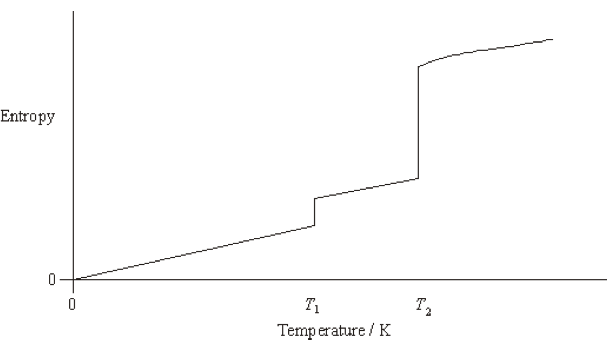
(source: http://satgamsat.blogspot.kr/2014/11/a2-chemistry-aqa-nelson-thornes-answers_9.html
_ looks like it was an SAT chemistry past problem lol)
You’ve seen this kind of graph a lot, right?
Probably you saw one back in middle/high school under the theme “water has a large specific heat”.
But it’ll be a bit different from that one.
This graph has temperature T on the x-axis and S on the y-axis,
and the graph you probably saw in middle/high school was probably this one
(if you learned entropy back in middle/high school, you might get hit with nuclear-grade chaos, right?)

This was it, right?!??!??!@@@
Well whatever, this one or that one, it’s the same story hahahaha hehe
Alright, let’s push on.
The entropy needed when a phase transition happens (in order to undergo the phase transition) is~~~??!?!!
I’ll just answer this question and move on.

Okay okay okay so what’s that, what is that
Let’s express our ΔS — at least roughly — in terms of existing physical quantities!!!!!
We’re going to infer ΔS very roughly, but I wouldn’t say this is completely meaningless.
Why????~?? That comes at the very end!!!
How exactly is it very rough??!?!?
Well
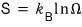
right???? So, it’s related to microstates.
Can we say this physical quantity Ω, the microstates, is proportional to volume V?
But, if there’s more than one, several of them,
particle 1:
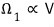
particle 2 as well:
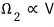
particle 3 as well:
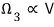
particle 4 as well:
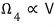
particle 5 as well:
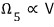
.
.
.
.
That is,

Now I’ll go into the equation trickery.

That is,
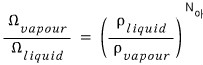
And among commonly known facts, there’s “the fact that the density of water is about 1000 times greater than the density of water vapor”.
Therefore
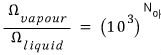
Got it up to here???
But why did we do this???
The goal was, when a phase transition happens, to express the change in entropy in physical quantities that are easy for us to handle.

So, the entropy change that happens when going through a phase transition from phase 1 to phase 2 is

This equation is called Trouton’s rule.
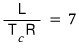
Can we say we’ve found some kind of universal law?
No, no, saying that would be too extreme.
It’s more like a very rough guess (?),
but as a ballpark estimate, it does hold.

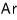
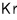
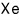

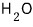
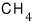
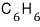
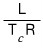
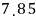
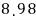
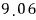
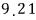
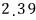
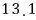
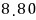
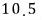
For a lot of particles, we can speak roughly like this.
Rou~ghly it all fits, but there are ones that clearly deviate!!!
For instance,
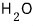
or

deviate a lot — why why where what is different about them that makes them deviate from Trouton’s rule??!??!
He is said to be because of quantum effects, and
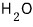
is said to be because of nothing other than polarity~~
Originally written in Korean on my Naver blog (2016-07). Translated to English for gdpark.blog.