The Clausius-Clapeyron Equation
We revisit phase transitions using Gibbs free energy and chemical potential to derive the phase coexistence condition and work toward the Clausius-Clapeyron equation hehehe.
We looked at phase transitions from the perspective of ’latent heat'.
This time let’s look at them from the perspective of free energy too
<actually, we’ve seen it before…. back at the van der Waals gas hehehehehehe>
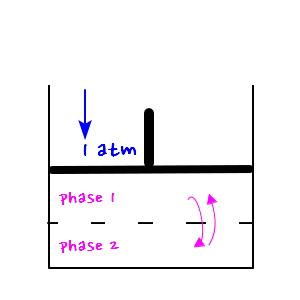
Since we’re considering phase transitions, I’ll sketch the diagram like this,
and given the situation we’re in now, you can sense that we shouldn’t use the expression from the Canonical ensemble, right hehehe
So I’m going to write Gibbs’ free energy like this.
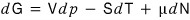
I’ll be using this kind of expression hehehe
Now then
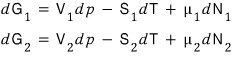
But, as you can see from the chemistry experiment above,
p can only be constant at atmospheric pressure,
and since we’re discussing phase transition, we’ll consider the temperature fixed at T_c.
Therefore
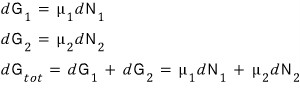
And let’s suppose we’ve reached equilibrium.
Equilibrium is the state of having reached minimum Gibbs energy!
Therefore it’ll be a state that doesn’t change at the minimum value of the Gibbs function.
That is,

Actually this is the kind of conclusion we’ve already drawn earlier,
“In equilibrium, ‘particles’ move from places where the chemical potential is high to where it’s low, and they move until the chemical potentials become equal.“
Now then, let me bring back the ‘phase coexistence line’ again.
(The part we’re studying now is ‘phase transitions’, and the phase coexistence line is the set that represents the moments ‘when the phase coexistence condition’ holds, where the phase transition takes place… so we can’t skip the discussion on the phase coexistence line)
In #57,
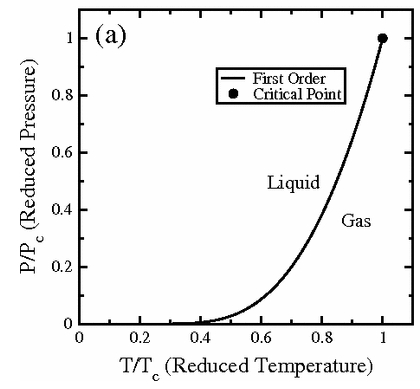
(Source: https://www.researchgate.net/figure/235737561_fig3_Figure-1aThe-vanderWaals-phase-diagram-Along-the-liquid-gas-equilibrium-line-a)
I showed this graph as the phase coexistence line for the van der Waals gas,
but this time
I guess I’ll just draw it myself.
First, I’ll swap the x and y axes. I won’t use reduced variables.
Then it’ll be drawn like this.
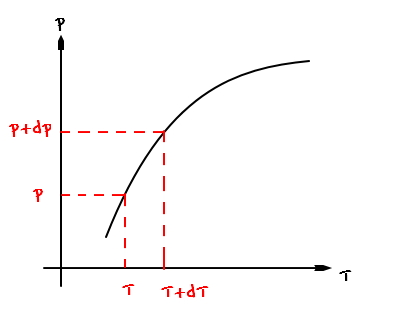
And I’ve tried marking two points,
and each of the two points would be representing this kind of situation.
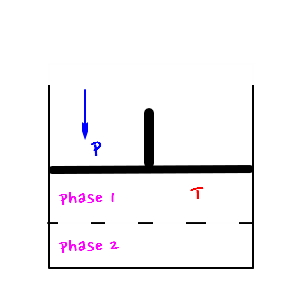
&&&
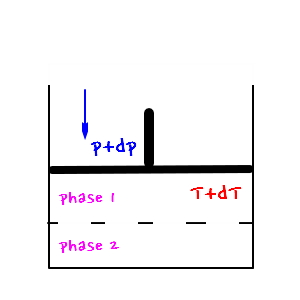
Both conditions are conditions that let the two phases coexist.
In both cases the reaction would have proceeded until each of the chemical potentials became equal
And the chemical potential was a function that depended on pressure and temperature like this.
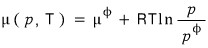
That is, in the case of the left side of the above figure it would be
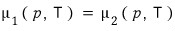
and
the figure on the right would be
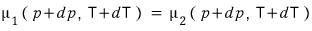
.
Now then,
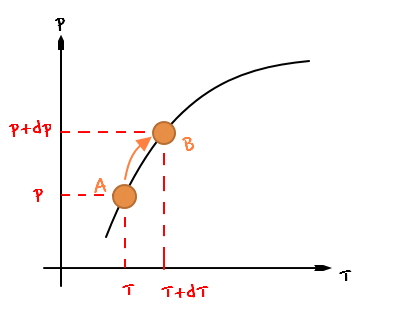
let’s think about changing from point A to point B (changing from the (p,T) situation to the (p+dp, T+dT) situation).
If p and T change, then μ, the dependent variable made from them, will also change.
But!!!!
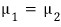
since we’re letting it change ‘along the phase coexistence line’ from the situation where this held

And G = μN, right??!?
Let’s read μ as the ‘Gibbs function per particle’
That is, saying that μ increases
can be read as: the Gibbs function per particle increases. So
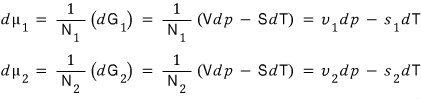
this is what’s the saaaame now~~~~~~~
Now then, since we said the two changes are equal
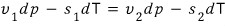
Let’s tidy up the equation a bit
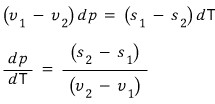
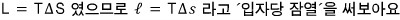
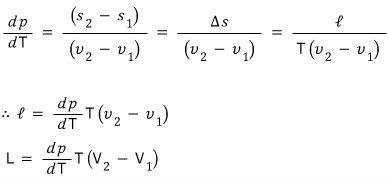
So the “(per particle) latent heat” is determined by the gradient of the p-T phase coexistence line, the temperature at that time, and the difference in the (per particle) volumes of the two phases :)
And~~~~~~~~~~~~~
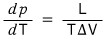
And crucially, this equation
is called the Clausius-Clapeyron equation,
and we’re going to be using this equation whenever we need an equation that means ‘phase coexistence’ from now on :))))))hehehehe
Sooo, what I’m going to do in the very next post is,
I’m going to derive the phase coexistence line again using the Clausius-Clapeyron equation hehehe so see you in the next post~:)))))))))
Originally written in Korean on my Naver blog (2016-07). Translated to English for gdpark.blog.