Temperature Dependence of Latent Heat for an Ideal Gas
We derive the liquid-gas phase boundary with the Clausius-Clapeyron equation, then dig into why latent heat L actually depends on temperature.
Example 28.2
Derive the equation for the phase boundary between liquid and gas, under the assumptions that the latent heat L is independent of temperature, the vapor is treated as an ideal gas, and V_vapour = V » V_liquid.
Let me solve this problem.
As I previewed earlier, I’ll solve the problem with the Clausius-Clapeyron equation.
The equation whose focus is ’the very moment a phase transition happens’ was the Clausius-Clapeyron equation,
and therefore the C-C equation can be understood as an equation about ‘phase transitions’.
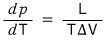
If we apply the ideal gas to this equation — since this is the equation for the phase boundary between the liquid and gas of an ideal gas —
applying the ideal gas equation (for 1 mol) pV = RT to the above equation,

But if we apply the ideal gas to this again,
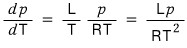
So then this would be the equation for the phase boundary.
(Of course, an ideal gas won’t undergo phase transitions, though.)
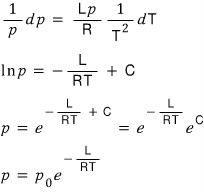
*****But, I had understood L as some ‘property’ that didn’t depend on anything,
but actually, L is said to be a physical quantity that depends on temperature.
Let’s have a look.
To figure out how L depends on temperature, what do we do!!!
Apparently there’s a sort of ewcipe (recipe).
Rather than just thinking of L as L(T)
and differentiating with respect to T, we’ll use L(T)/T!!!!!!! And through that we’ll look at L’s temperature dependence. (Like entropy?!)
So if we simply differentiate L(T)/T,
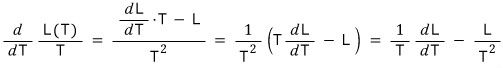
But, we can also pull out an equation like this.
We use L = TΔS.
In this case, it would be L = T(S_vapour - S_liquid).
Then get a feel that L/T can equivalently be expressed as ΔS,
and we separate the change of L/T into a term from p and a term from T.
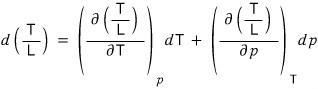
Now that we’ve written it like this, let’s get into the shenanigans.
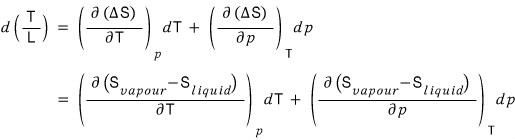
What we need to find is
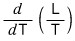
this, so
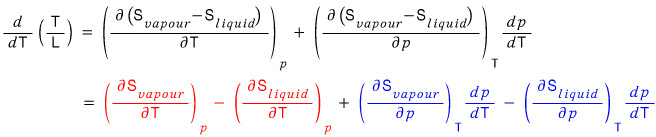
Organizing it like this,
for the red part we’ll use TdS = dQ,
and the blue part we’ll organize using Maxwell’s relations.
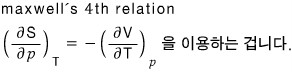
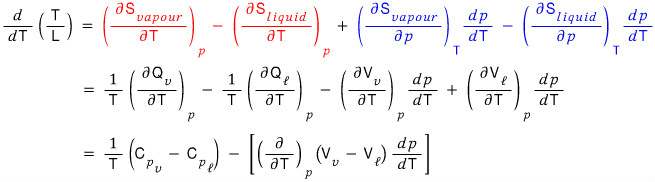
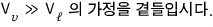
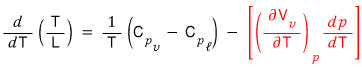
For the red part we’re going to apply the ideal gas equation of state,
first, dV/dT = R/p,
and for the latter term, we can just apply the Clausius-Clapeyron equation with the ideal gas equation applied that we did waaaay up there~ yup~
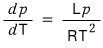
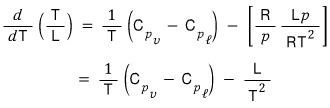
Okay okay okay so now
we’ve got two expressions for d/dT(T/L).
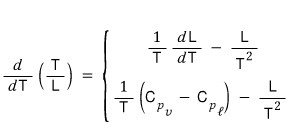
Therefore,
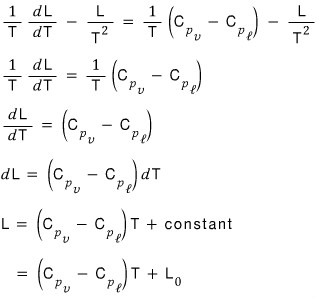
So the phase coexistence equation we made earlier with the ideal gas assumption thrown in was
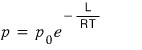
rather than this,
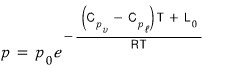
this is the rigorous equation!!!
But the point here was to realize that “actually, the latent heat L depends on temperature!!!!”
However, in the discussions that follow, we’ll keep saying “yeah we know it’s temperature-dependent, but let’s just ignore it for now~~~”
and carry the logic forward that way hhh hehehehe
Alright, bb
Originally written in Korean on my Naver blog (2016-07). Translated to English for gdpark.blog.