Phase Coexistence Curves of Water
We draw out phase coexistence curves using the Clausius-Clapeyron equation and figure out why water's solid-liquid line has a negative slope — spoiler: hydrogen bonding!!
Alright alright alright anyways so the takeaway from what we’ve been discussing up to now is
“the equation for the phase coexistence line = the Clausius-Clapeyron equation”. And yeah.
Now let’s draw it!!!! hehehehe

Let’s just draw it roughly like a log function
Then it would look like this.
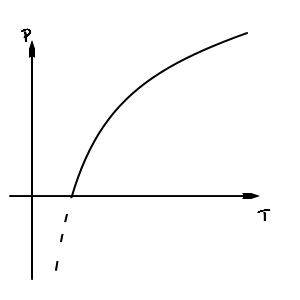
Something like this, right
Okay so let’s think of this picture as the phase coexistence line derived from the Clausius-Clapeyron equation between vapour and liquid
and let’s derive the phase coexistence line between solid and liquid.
The equation here will be the same as above too,
but this part will be different.
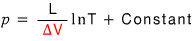
Forget about L, let’s look at V
This delta value will differ depending on the substance, but ΔV has something in common.
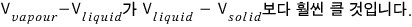
This point right here….hehehe
So then the log function between liquid and solid will be steeper than the log function between vapour and liquid.
Then I guess we can just overlay it like this and draw it.
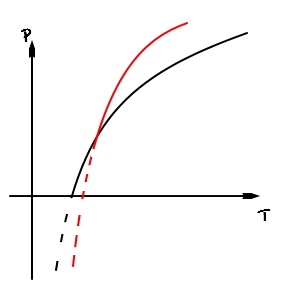
Ah, then what we can confirm here is ’the existence of the triple point’ — looks like we can find this too
(it’s a pretty qualitative analysis though;;;hehehe)
This posting was actually for the sake of talking about what comes below.
Actually for the case of water, you also learn this in middle/high school, but the phase coexistence line looks like this hehehe
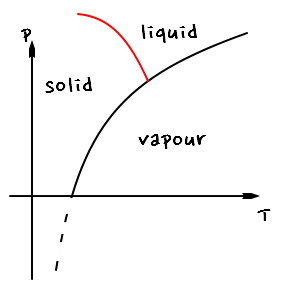
It’s like this. The point is that the gradient of the phase coexistence line between solid and liquid is (-)
What it being negative means is
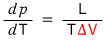
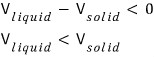
that’s what it means.
That’s really weird, isn’t it
If we represent solid, liquid, gas simply,
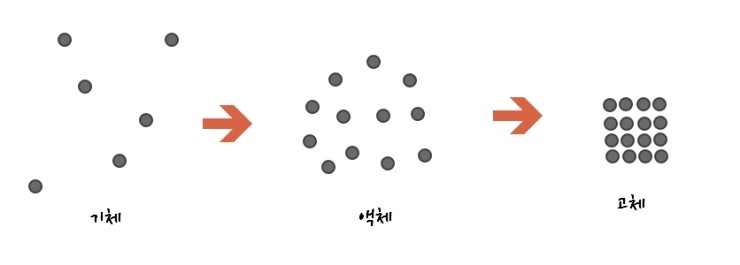
this seems reasonable, right?!?!?!?!? But,
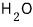
in the case of
is what it ends up saying
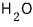
why does it have this kind of property?????
We learned this in high school chemistry, right?!?!?!? It’s hydrogen bonding!!!
FON soo FON soo ~~~~~~ that’s how I memorized it lolol
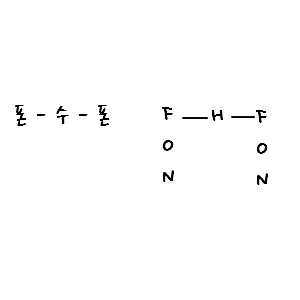
Let me try interpreting “low temperature” as “low contained energy”
(things that can be contained energy: translational kinetic energy, rotation, vibration, etc.)
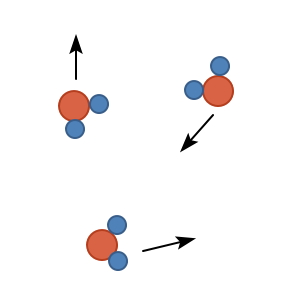
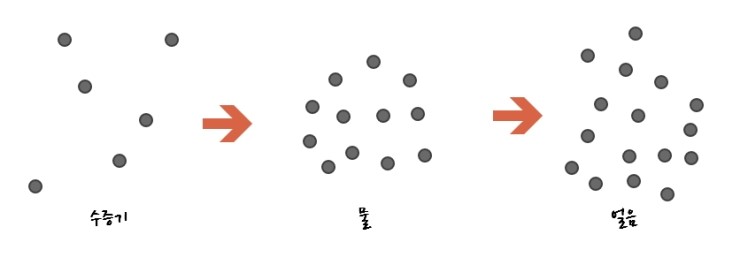
Water molecules at high temperature, i.e. water vapour, have so much contained energy that
as in the picture above you could say they’re thrashing around like hell.
Then at low temperatures

since the contained energy is low
they don’t move, don’t rotate, don’t vibrate, right?????
A little bit right.
It’s true that they don’t move, but it’s not like they sit still like that.
These guys go find their own spots.
Because the difference in electronegativity between H and O is super big
ummm~~~~ the electronegativity of O is so big that it pulls in the (-) influence of H, so water molecules end up with polarity.
So it’s not that they’re huddled together haphazardly like above
they line up in rows and columns.
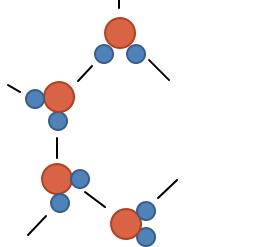
They end up lined up in rows and columns like this
and if the temperature gets juuuust a little bit higher than this
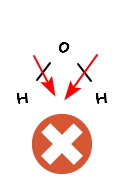
rather than the thermal energy dissociating the water molecules like this
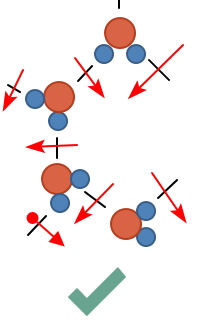
since the thermal energy is used to break the hydrogen bonds like this
so compared to when it was ice, the liquid ends up in a juuust a little~ more aggregated state hehehehe
This isn’t chemistry class, so I won’t get into stuff like how 4℃ water has the largest density that
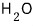
can have, and so on.
Just understand the phenomenon that differs from ordinary particles due to this hydrogen bonding,
and that glaciers float on water, and the Han River freezes from the surface down, and stuff like that…heh.hehehehe
Pleeease look forward to the next posting hehehe
It’s gonna be really fun heh
Originally written in Korean on my Naver blog (2016-07). Translated to English for gdpark.blog.