Kelvin's Formula and Why Water Boils
We dig into why water boils by layering surface tension on top of phase-transition theory, then crank through the math to land on Kelvin's formula.
Alright, this time we’re going to analyze another phenomenon.
Why water bubbles and boils!!!!! Then let’s begin.
Earlier, you see.
For phase transitions (van der Waals gas), we drew the Gibbs function like this.
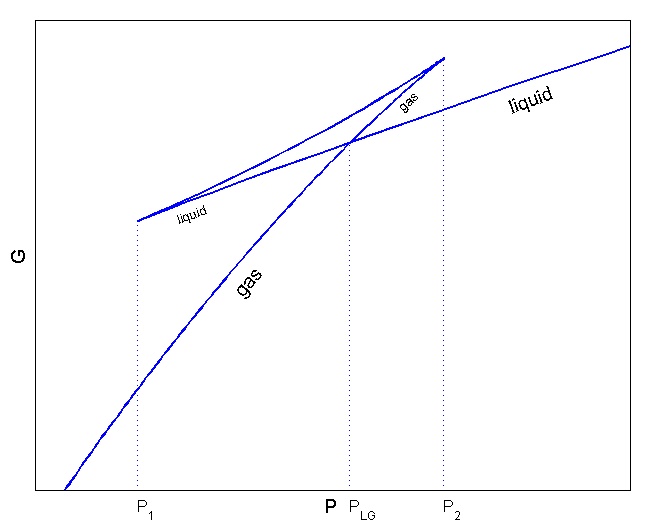
(Source: https://commons.wikimedia.org/wiki/File:Van_der_Waals_GibbsP.png)
Now then, between the chemical potential μ and G there’s the relation

, so
if we now change the graph above to a ‘per particle’ basis,
we could draw it like this.
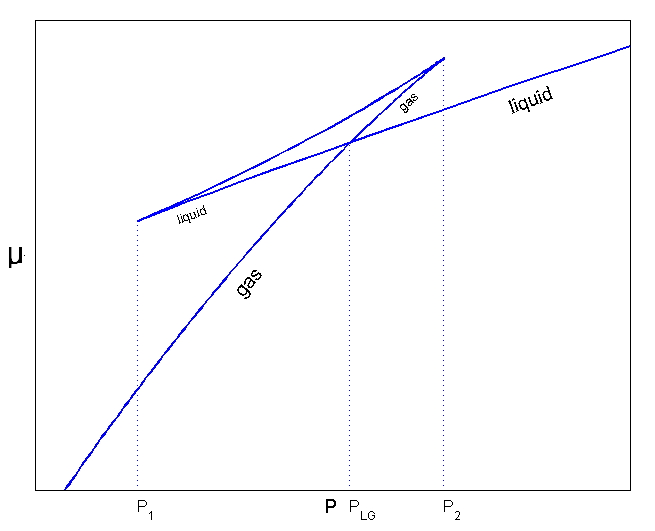
I just switched the y-axis to the chemical potential μ.
As we dealt with before, this graph raises the possibility of the existence of superheated liquid and supercooled vapour,
and actually explained those phenomena!!!! (That was #56.)
But you see. This time I want to take one more step forward.
What I’m going to consider further is “surface tension”.
So let me quickly touch on surface tension.
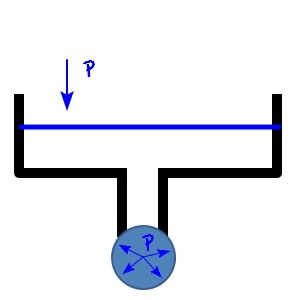
If we hang a liquid on a piston shaped like this, a droplet of liquid will form as in this picture.
The reason liquid forms into droplets is the attractive force between the molecules that make up the liquid.
Let’s say the surface area of the water droplet above is A.
The surface area is A, and if on top of that we consider the surface tension,
that is, if we weren’t considering surface tension (only attraction) and then suddenly consider it,
it amounts to considering ‘more’ of the attractive force between the molecules that make up the liquid.
Then the surface area becomes ‘smaller’ by that much more consideration,
so we just have to look at how much smaller the surface area gets.
Let’s call the work needed to make the surface area smaller by that much dW, and
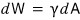
let’s write it like this.
γ is some constant that determines the surface tension for each substance~~~
Now,
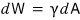
let’s express this in other things that are useful for us to use.
First, since dW is the amount of work ‘done’,
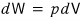
.
And if we think of the water droplet as a ‘sphere’,
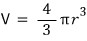
, so
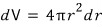
Then,
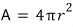
, so
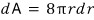
Wheee, then we can reconstruct the surface tension equation.
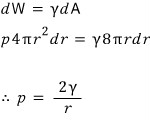
.
So now let’s get back to the main topic, phase transitions,
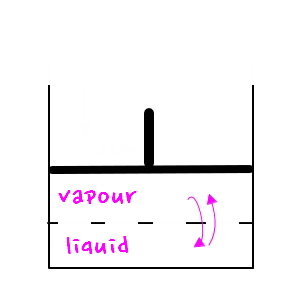
let’s say we’re in equilibrium right now.
If we interpret “equilibrium” as “it reacts until μ becomes equal”,
then because we’re in equilibrium, liqi
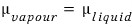
, we can say it’s in this state.
And let’s say the current equilibrium is
the state where the chemical potentials are equal at this point.
Right now vapour and liquid are coexisting, right?!?!?!
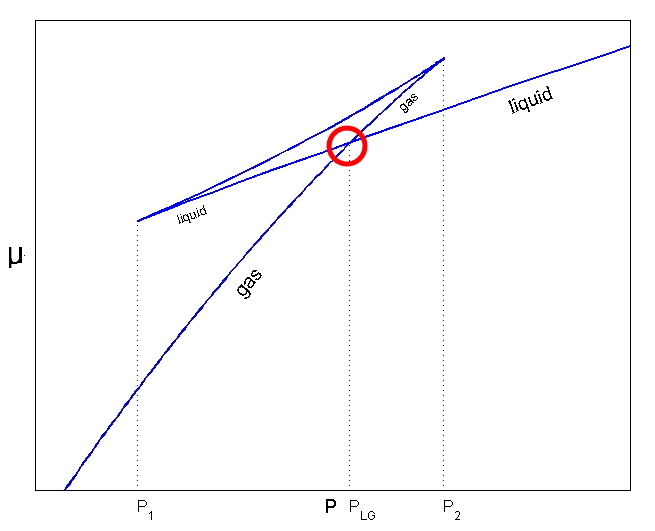
In this kind of situation, sud.den.ly !!! let’s say we consider the surface tension γ.
In a situation like the one above, let’s say we sud.den.ly consider the surface tension gamma.
Then, the pressure of the liquid will change ever so slightly.
It used to be that p, and it changed ever so slightly, by dp_liquid, so the pressure became p+dp_liquid.
Whaaat??!?! Then what happens to the reaction?!?!??! The reaction continues until μ becomes equal??!?!!
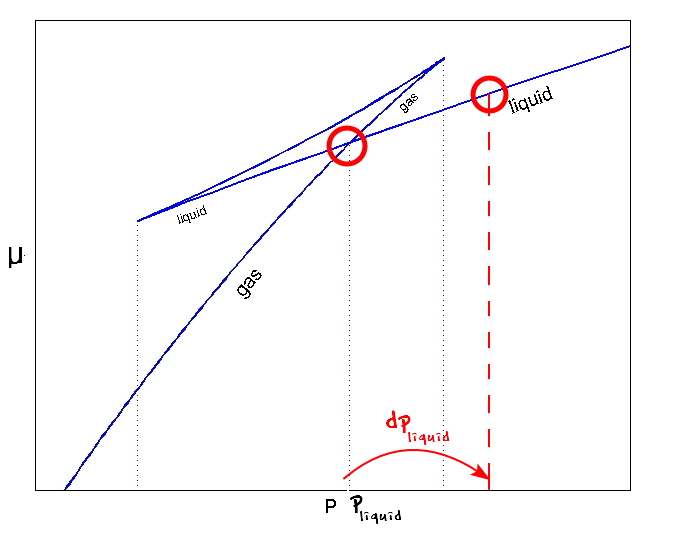
Aha~~~μ_vapour also changes!!!
It will continue to react until it becomes equal to the chemical potential of the new liquid.
Then how much does the vapour change by,
it’ll be this much.
Aha~~~~ μ_vapour also changes!!!!
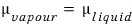
is matched again!!!
But if you look at the graph, p_vapour changes, but it doesn’t change by dp_liquid
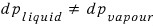
it’ll be
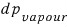
the magnitude of it is shown in the picture above.
Now let’s go fully into graph analysis.
Right now
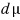
are equal to each other
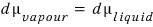
the magnitudes are equal to each other, but let’s express dμ ‘differently’
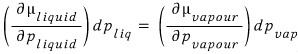
We can write this equation, and the principle is
. And if we let the pressure of the piston be p,
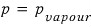
it will be, so I’ll rewrite the equation above like this
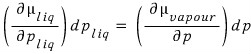
Now, we had dG = -SdT + Vdp.
So
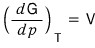
it is, and since μ is the “Gibbs function per particle"
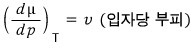
(It’s not the volume of the particle, but the volume per particle occupied in the gas, liquid, or solid state.)
So the equation above becomes
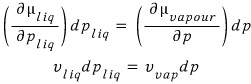
And if we multiply both sides by Avogadro’s number N_A,
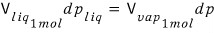
Here the volume now becomes the volume occupied by 1 mol of particles.
Now, let’s assume V_vap is an ideal gas. (It’s just assuming the gases are ideal gases.)
Then, since the volume of 1 mol of ideal gas is RT/p,
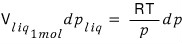
Now we integrate both sides.

What was the dp_liquid that we originally considered?!??!!
It was the magnitude of the pressure change in the liquid due to surface tension.
Then that p_liquid is
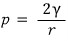
is what it’ll be
We considered this much of a magnitude sud.den.ly.
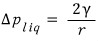
that’s the gist,
let me put it like this.

You get it, right
This equation is called Kelvin’s formular~~~hehe
Now then let’s try to understand that equation.
Right now we
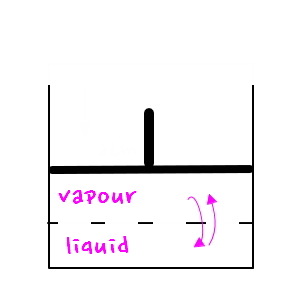
understood it with this picture, but since we’re going to understand a natural phenomenon, I’ll manipulate the picture just a little.
I was going to say let’s manipulate it like this, but I failed to express it with MS Paint.
Rather, at least for this picture, a hand-drawn one would be better.
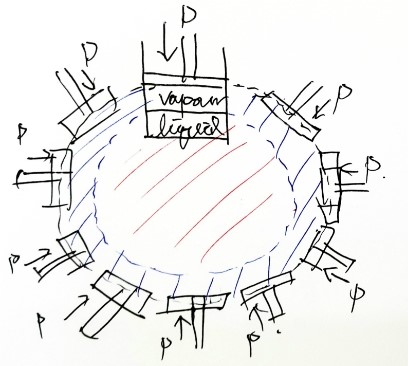
I want to draw it like this~~~ let’s think of it as a rotated picture.
It’s all the same thing anyway, right????
So right now
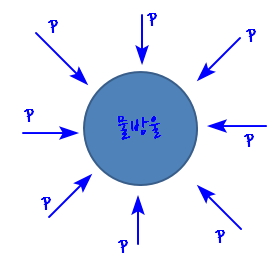
I’m saying let’s think of it as a water droplet.
And Kelvin’s formular is
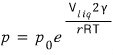
this, and p is the vapour pressure, which is the pressure of the vapours on the outside.
Now then what this equation says is
that the smaller r is, the bigger p gets.
That is, p getting bigger means the tendency to evaporate gets bigger
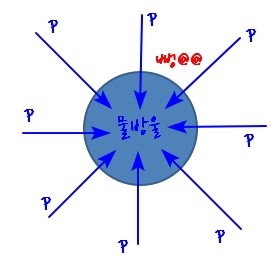
You can just imagine it going pop@@@ and bursting.
(I drew the direction of p from outside to inside, but I think it’s fine to imagine the picture with the direction of p set from inside to outside the other way.)
This is saying that because of the tendency to evaporate, supercooled vapour can exist.
That is, you could say that depending on the γ value determined by the substance’s properties, it determines how small a droplet has to be to go pop@@@ and burst,
and also, even when the temperature in the atmosphere is low enough for water droplets to form,
because of the tendency for small droplets to evaporate, they can remain in the water vapour state!!!!
Anyway, k cool?!?!?
So then what do we have to do for condensation to happen in the sky?!?!
If droplets with a large radius have to form?!?!?
If only small droplets form, it doesn’t rain,
so you sprinkle silver iodide or dry ice, things harmless to humans, into the sky
to get water to condense in the sky!!!~~

For this kind of purpose..
Then, by creating the possibility for larger droplets than before to form, it makes droplets form, apparently.
We can also think about the opposite case.
Just a moment ago we were thinking like this, right?!?!?!?

The opposite case is

The surface tension of the liquid will pull it more outward like this.
That is,
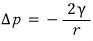
we’re ‘additionally’ considering.
Therefore
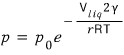
Now then, a water vapour bubble formed inside the liquid!!! If its radius is small, now p becomes smaller than before.
So this means that even a gas at a sufficiently high temperature can exist as a superheated liquid.
For a bubble to form in this situation?!?!?!? The bubble has to be big enough.
That is, among the many bubbles generated inside the liquid, the small bubbles all croak, and only the big bubbles survive and keep growing and growing, and th
at’s what appears to our eyes as “water bubbling and boiling.”!!!!!
(The reason they keep growing and growing will be revealed in the next post. Continuously growing is the natural thing.)
Then you can also understand why when you put ramen seasoning into boiling bubbling water, it goes quiet again
and only starts bubbling and boiling again after reaching a higher temperature.
The ramen seasoning that went in played the role of the ’nucleus’ for generating “small bubbles”!!!!!
Ah….. so “boiling point elevation” can also be seen this way……hehehk
Originally written in Korean on my Naver blog (2016-07). Translated to English for gdpark.blog.