Gibbs Free Energy of a Water Droplet and Bubble Nucleation
Walk through calculating the Gibbs free energy of a liquid droplet in vapor equilibrium, factoring in surface tension to set up the physics behind bubble nucleation.
Example. 28.6
Calculate the Gibbs function of a liquid droplet of radius r that is in equilibrium with vapor.
Assume the temperature is the temperature at which the liquid is in a thermodynamically stable phase.
Since we just said “in equilibrium,” let’s say everything is constant.
Then p, T will also be constant and therefore μ is constant as well.
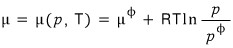
So, since G = μN,
we can write dG = μdN,
And since we’re considering vapour and liquid
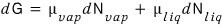
let’s write it like that.
But, since we’ll also consider surface tension,
we add the term due to surface tension onto the above Gibbs function.
Since dW = γdA,
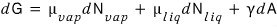
And the law of conservation of particle number
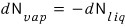
Therefore
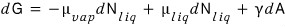
it’s fiiine to do it like this~
Also let’s include the relation we used earlier
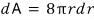
as well.
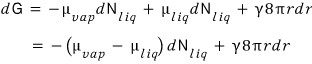
Now, we’re going to express dN in terms of r,
let’s think about it like this.
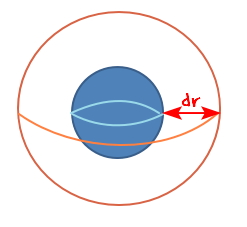
If the radius of the droplet grows by dr, the volume grows by
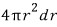
this much, right?
How many liquid water molecules are in this much of added volume???
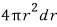
the mass of the liquid with volume
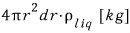
is this.
And if we divide by m, the mass of a single liquid molecule, we’ll get the number!!!
That is,
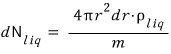
Therefore
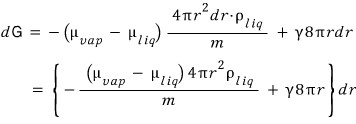
Now if we bash an integral on both sides
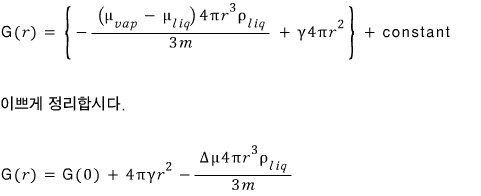
We’ve derived the Gibbs function for a droplet of radius r.
Let’s try plotting it
Ahhhhh right

Now shall we really plot it
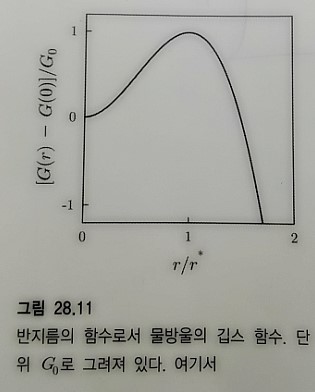
I’ll just copy the one in the book.
Well then.
Let me try computing the extremum of the above graph where dG = 0.
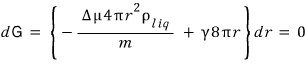
Since that’s what we’re finding,
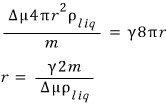
What this means is it maintains equilibrium at around that r.
Let’s call that r r_star.
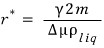
So, among droplets that form, one that forms smaller than this much reacts in the direction of Gibbs function minimization, so
r→0
“it disappears.”
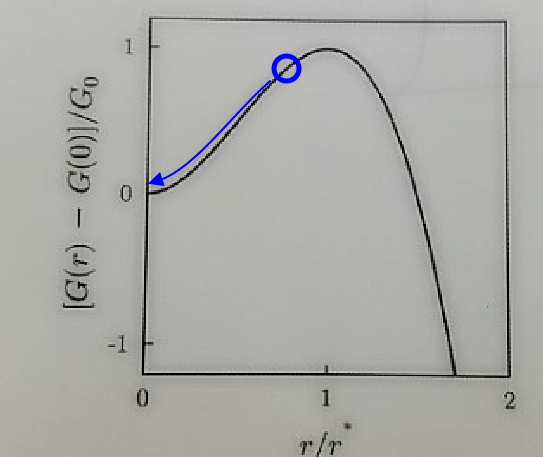
And also a droplet that forms bigger than r*, since it reacts in the direction of Gibbs function minimization,
r → ∞
“the droplet grows without bound.”
So from the previous posting,
when water bubbles and boils, the fact that the droplets grow
can be explained by the principle of Gibbs function minimization, it seems~ hehehe hahahahahahaha
Originally written in Korean on my Naver blog (2016-07). Translated to English for gdpark.blog.