Ebullioscopic and Cryoscopic Constants
Let's derive the boiling point elevation and freezing point depression constants from scratch using chemical potential and Raoult's law.
Let’s derive the boiling point elevation constant and the freezing point depression constant.
First,
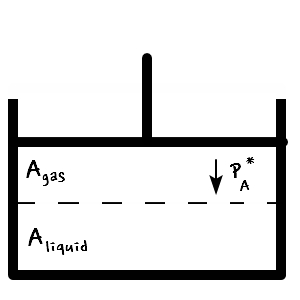
let’s imagine this kind of “equilibrium” situation.
Since it’s in equilibrium,
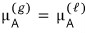
we can write this.
But right now, since there is only pure A, in the sense of “when there is only A”,
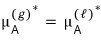
I’ll attach a star like this.
It means pure A.
And
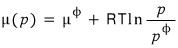
according to this relation, the above equation
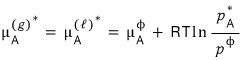
could be written like this without any problem.
But now in exactly that situation,
let’s say we injected another substance, B molecules, into the liquid!!!!
First, I don’t really know about the gas side. The pressure of A on the gas side was
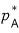
but I don’t know anymore.
First, since it’s clearly not pure A, let’s drop the star.
That is,
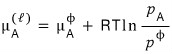
let’s write μ like this. I don’t know if it has changed (yet).
But since the reaction proceeds until μ becomes equal, the gas side would also have become equal to that value.
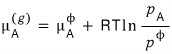
Now I’ll line up the two equations.
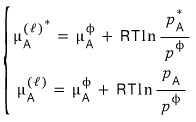
I’ll subtract the two equations. I mean, solve them as a system.
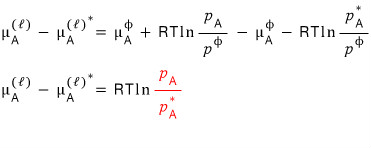
The red value above is clearly less than 1.
It has to be, because of Raoult’s law.
Raoult’s law is at the middle/high school chemistry level, so I’ll skip the detailed discussion.
Summary: the law that the vapor pressure lowering of a solution in which a non-volatile, non-electrolyte solute is dissolved is proportional to the mole fraction of the solute.
A law discovered through experiments in 1888 by the French physical chemist F.M. Raoult. In a solution of a non-volatile substance, the vapor pressure of the solvent in the solution is proportional to the mole fraction of the solvent, and the rate of vapor pressure lowering of the solvent equals the mole fraction of the solute.
For example, the vapor pressure of a solution of volatile ethanol dissolved in water is higher than the vapor pressure of water. Because ethanol has a lower boiling point than water and evaporates easily, it doesn’t hinder the evaporation of water and vaporizes faster than that. However, in a solution of non-volatile sugar dissolved in water, because the sugar molecules partially hinder the water’s evaporation, the vapor pressure of sugar water becomes lower than that of water. This kind of phenomenon is called “Raoult’s law”.
[Naver Knowledge Encyclopedia] Raoult’s law (Doosan Encyclopedia)
(source: http://terms.naver.com/entry.nhn?docId=1086837&cid=40942&categoryId=32252 )
So
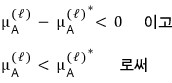
we can conclude “when mixing happens, μ gets smaller”.
Now bringing the above equation back, let’s set the “mole fraction” as
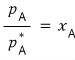
.
Then
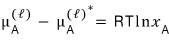
after writing this, I’ll play around with it a bit.
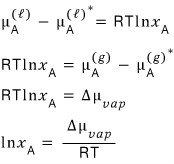
Chemists apparently call the chemical potential μ the “Gibbs function per mole”. (Whereas physics people call it the Gibbs function per particle….)
So with just a little scaling, converting that value to “per mole” would be no work at all.
So I’ll just write it like this.
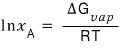
But, G = H - TS.
And also right now it’s in a constant equilibrium state with p and T*,
that is
dG = dH - TdS
Therefore
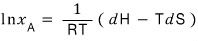
And
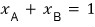
since this,
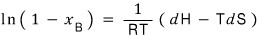
let’s write it.
Now let’s approximate the above equation by doing a Taylor series expansion.
Doing so,
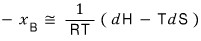
dS = dQ/T, and since p is constant right now,
we can write dQ = dH.
That is dS = dH/T*
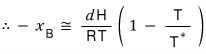

Right now the left-hand side means “the amount by which” the temperature at which the phase transition occurs has changed.
In chemistry, there is the proportionality that the change in boiling point is proportional to the molality,
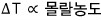
there’s this proportional relation,
that is
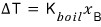
it has to be expressed like this, you see?????
But, right now we
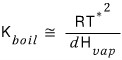
have found this.
This is called the boiling point elevation constant (ebullioscopic constant)
and by the same principle,
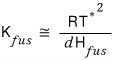
the freezing point depression constant (cryoscopic constant) can also be derived like this, apparently.
And we should point out that the dH here means the amount of “latent heat”!!!
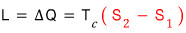
it was, and since p is constant dQ = dH, and
so…. well anyway, so we can also say that the red part makes sense in that as solute B comes in, the entropy becomes more and more and more complicated…. right?!?!?!? hohohoho lol
Originally written in Korean on my Naver blog (2016-07). Translated to English for gdpark.blog.