Symmetry Breaking
Turns out not all phase transitions are the same — Ehrenfest classified them by order, and second-order ones like superconductivity behave totally differently!
This post goes beyond the undergraduate level. So the main purpose of this posting is “let’s at least hear about it once.”
We’ve been learning about phase transitions,
and every time we say ‘phase transition,’ we naturally thought of liquefaction, vaporization, melting. . . . things like that
Okay so
Other than these phase transitions, what other phase transitions are there????
Mental breakdown?!?!?!?? lollllllll
No No, there are other phase transitions we know
As mentioned earlier, there’s “superconductivity” or “superfluidity”
You’ve heard of them, and you know what kind of phenomena they are, right????
Then would the superconducting phase transition and the solid-liquid-gas phase transition have the same process????
Feels like not, right???? I’m only asking this way because they aren’t lolll
Yeah, they’re not. They have different processes
Huh?!????? How are they different????
Paul Ehrenfest classified these, apparently.
If you draw the T-G function graph for the phase transition between solid-liquid-gas,
what did it look like, it was like this
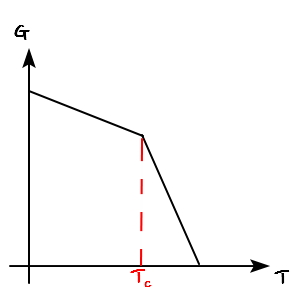
We learned earlier that the Gibbs function for the solid-liquid-gas phase transition bends.
And, dG = -SdT - Vdp.
Ohh let me draw the first derivative of the G-T graph.
Eh, why do I say first derivative so grandly
I just need to draw the S-T graph.
We drew this earlier too.
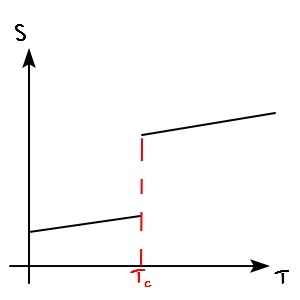
It looked like this, and that jump interval there was called the “latent heat”
Alright, let’s draw the second derivative of G too.
I just need to draw the first derivative of the S-T graph
wait a sec~
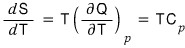
Ah~~~so the second derivative of G was C_p.
For this, rather than me drawing by hand, attaching an experimental result from google will probably have a better effect on credibility!!!
It looks like this!!!!!
Alright, Ehrenfest named the case where G and the first and second derivatives of G have this kind of pattern
“first-order phase transition.”
Now then, here’s the thing.
They’re still phase transitions, but there are ones where G, G’, G’’ show a different pattern from this.
There are things that undergo phase transitions showing a different appearance from first order phase transitions
2nd, 3rd, 4th,…. there are many, apparently.
Alright, so let me show an example, a second-order phase transition
By showing a first order phase transition and a second order phase transition at the same time,
I’ll show what the differences are.
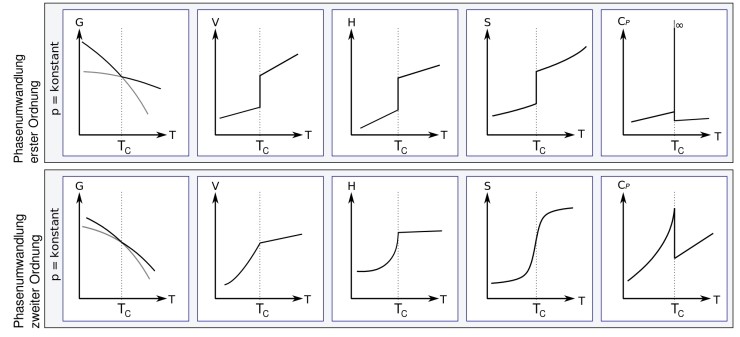
The first row is the first order phase transition we saw above,
and the second row shows the various derivatives of the G function during a second order phase transition.
Superconductivity and superfluidity are included in this second-order phase transition category.
Up to here was the late 19C ~ early 20C, apparently.
But, there’s a fundamental problem here.
Phase transitions occur at the critical point,
and at that critical point, there are enormous fluctuations in the system beyond imagination
What kind of error does these enormous fluctuations bring,
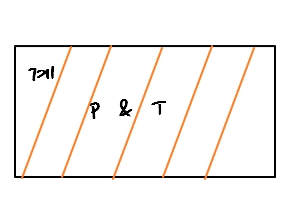
we based things on pressure, or temperature T, etc., being uniform or in equilibrium across the entire system.
But, if there are enormous fluctuations.
That’s honestly absurd
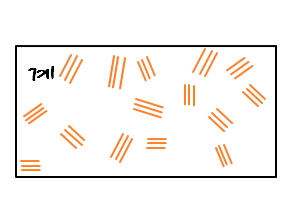
in this kind of way, the state functions we dealt with before become non-uniform at each and every point,
so Ehrenfest’s classification like that is too irresponsible, isn’t it….
<at the place where the phase transition occurs, it doesn’t follow the analysis we expected>
That kind of opinion was dominant, apparently.
So in modern times, when classifying phase transitions,
whether it includes latent heat or not,
first-order phase transitions that include latent heat, and Ehrenfest’s 2nd, 3rd, 4th, etc. ‘continuous phase transitions’ that do not include latent heat
they only distinguish up to about this level, apparently.
So
scientists, when discussing phase transitions, paid attention to “symmetry breaking” rather than the shape of the Gibbs function, apparently.
This is a very broad concept, but because it can talk about phase transitions in general, encompassing everything,
it’s expected to present us with a new paradigm for phase transitions
So apparently, that’s its significance
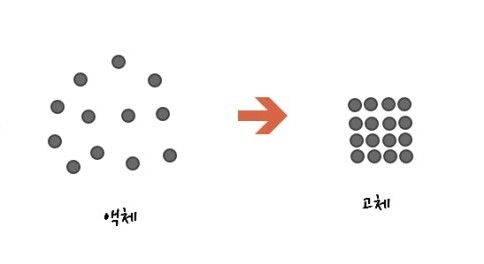
Let’s take a look at symmetry breaking.
Look at the picture above and think about which is more symmetric!!!
The answer came right away
At first you’ll think the solid is more symmetric. However, the correct answer is….
the more disordered liquid is more symmetric, apparently.
Because whether you view the liquid after moving it a little, or after rotating it (turning it) a little, it’ll all look the same.
On the other hand, for the solid, if you move it a little and look, it’ll look different, and even if you rotate it, it’s different from before.
I said the concept of symmetry breaking is broad. Not only solid-liquid-gas phase transitions but symmetry breaking can also be applied to other phase transitions, apparently.
For example, between ferromagnetism ↔ paramagnetism, you can see spin symmetry breaking,
and in the phase transition between normal metal ↔ superconducting metal, you can see wavefunction symmetry breaking, apparently.
big bang!!!! early 10^-43 seconds
So, around 0.000000000000000000000000000000000000001 seconds, universal gravitation was born, and the next moment electromagnetic force, weak force, strong force were born, right?????
This is also apparently because as the universe briefly expanded and the universe cooled, they were born in order via symmetry breaking following the Higgs mechanism…
For me it still doesn’t make sense,,,, hehehe
Anyway, this is saying that concepts like symmetry breaking, phase transition, critical point are important.
As for the details, I’ll study them in grad school…. will I study this kind of thing? lolllllllllll I should probably just buy a book and read it loll
Later when I’ve organized it, I’ll upload it as a fun posting heheh
Originally written in Korean on my Naver blog (2016-07). Translated to English for gdpark.blog.