Practice Problems: Chapters 26 & 27
Working through practice problems from Chapters 26 & 27 on van der Waals gases, isothermal compressibility, and isobaric expansivity near the critical point — 27.2 was already covered, so we're skipping ahead~
Prob 26.1
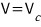
Show that the isothermal compressibility of a van der Waals gas can be written as
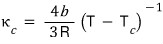
.
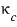
Sketch the temperature dependence of , and explain what happens to the properties of the gas as the temperature is lowered through the critical temperature.

Prob 26.2
The equation of state of a certain gas is
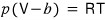
.
Here b is a constant.
What order of magnitude do you expect for b?
Show that the internal energy of this gas is a function of temperature only.

Prob 26.4 Show that the isobaric expansivity of a van der Waals gas
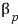
is given by
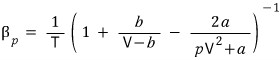
.
What happens to this quantity near the critical point?

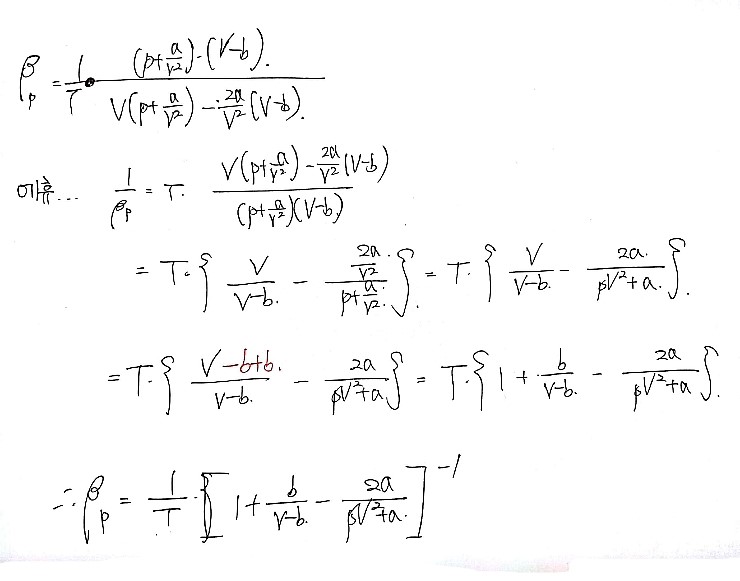

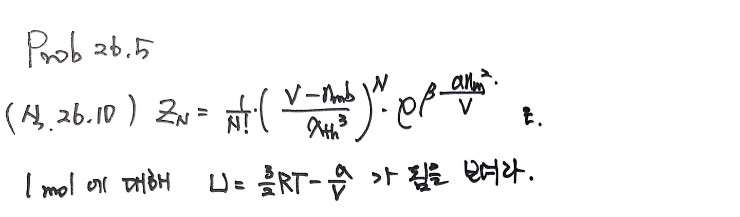

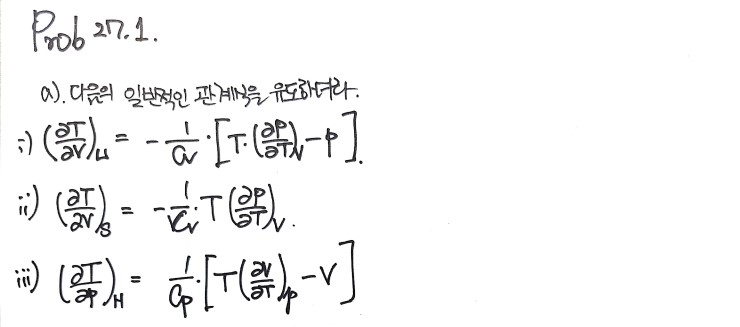



27.2 was something I naturally derived when covering the concepts, so it’s recorded already~
Originally written in Korean on my Naver blog (2017-01). Translated to English for gdpark.blog.